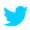
Permits from other national agencies are necessary for importing masks while mask export ban is lifted.
The COVID-19 pandemic has raged on the whole world and humans have been suffering since December of 2019. Masks are one of the essential and important shields against coronavirus. However, masks without official permits may harm rather than protect people’s health.
Therefore, importers or inbound passengers are required to apply prior approval from Food and Drug Administration, Ministry of Health and Welfare (FDA) or Bureau of Foreign Trade, Ministry of Economic Affairs (BOFT).
On the other hand, domestic mask supply has been enough and the remaining has been open to export from June 1, 2020.
-
Luggages of Inbound Passengers
-
Total under 250 pieces (medical and non-medical masks): bypass
-
Total over 250 pieces: Permit of BOFT
-
Medical Masks over 250 pieces: Permit of FDA
-
-
Imported Cargo
-
Medical Masks:
-
Permit of BOFT
-
Permit of FDA
-
Inspection by FDA (exception: personal use)
-
-
Non-Medical Masks:
-
Permit of BOFT
-
-
Additionally, both FDA and BOFT provide online application systems: FDA、BOFT
The mentioned regulations may be amended depend on the domestic demand or other reasons, so please be alert to the media release on Customs Website.